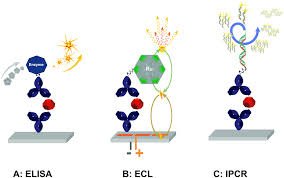
ADA Assay Performance Monitoring During Long Clinical Trials
Consistent and reliable data are critical to the success of long-term clinical trials. Maintaining the integrity of bioanalytical methods over multiple years presents a significant challenge, particularly for immunogenicity assessments. Monitoring the performance of an ADA assay is essential to ensure that results remain comparable from the first to the final sample. A structured monitoring strategy prevents assay drift, safeguards data quality, and ensures that immunogenicity measurements accurately reflect the patient’s immune response throughout the study, supporting robust regulatory submissions.
Establishing a Reliable Baseline
The success of a multi-year study begins long before the first sample is analyzed. It starts with robust ADA assay development and validation. A well-validated assay provides the baseline performance metrics against which all future runs are measured.
During this initial phase, scientists must define critical parameters:
- Cut-point determination: Establishing a statistically sound cut-point that minimizes false negatives while managing the false-positive rate.
- Sensitivity and drug tolerance: confirming the assay can detect low levels of antibodies even in the presence of the drug.
- Selectivity and specificity: Ensuring the assay distinguishes between the ADA antidrug antibody and other matrix components.
A solid baseline allows the bioanalytical team to detect subtle shifts in assay performance later in the trial. This is particularly important for complex methods like an NAb assay (neutralizing antibody assay), where cell-based variability can influence results.
Tracking Assay Drift and Variability
In an ELISA lab, variability is inevitable. Reagents degrade, equipment requires calibration, and analysts change. The goal is not to eliminate all variability but to control and monitor it.
Strategies for tracking performance include:
- Trending of Quality Controls (QCs): Plotting QC data over time (Levy-Jennings charts) helps identify systematic drift. If the low positive control consistently trends upward, the assay sensitivity may be changing.
- Plate-to-plate analysis: Monitoring the signal-to-noise ratio across plates ensures that background noise does not obscure true positive signals.
- Analyst performance: regularly reviewing data to ensure that technique differences between analysts do not introduce bias.
Regular review of these metrics allows for early intervention. If a shift is detected, the laboratory can investigate the root cause, such as a degrading reagent or a pipette out of calibration, before it invalidates a large batch of clinical samples.
Managing Reagent Lot Changes
One of the most common sources of variability in long-term ADA analysis is changes in critical reagent lots. Antibodies, plates, and biological matrices vary between manufacturing lots. In a trial lasting several years, utilizing a single lot of reagents is often impossible. To manage this, laboratories must implement a strict bridging strategy. Before a new lot is introduced, it must be tested alongside the current lot. The acceptance criteria should be predefined to ensure that the new lot yields comparable sensitivity and drug tolerance.
This process applies to all components, including:
- Capture and detection antibodies
- Blocking buffers and diluents
- Positive control antibodies
- Cell lines used in neutralizing assays
Failure to bridge reagents properly can result in a "step change" in the data, where the incidence of immunogenicity appears to increase or decrease solely due to the reagents used, rather than a biological change in the patient population.
Streamlining Data with Multiplexed ELISA
Efficiency becomes critical as sample volumes grow. Multiplexed ELISA and advanced ELISA analysis platforms enable simultaneous detection of multiple analytes. This approach is valuable when small-molecule bioanalysis is conducted alongside immunogenicity testing.
Multiplexing offers several advantages for long-term monitoring:
- Reduced sample volume: Conserving patient samples is vital, especially when multiple reassays are required.
- Higher throughput: analyzing more samples per run reduces the total time required for data generation.
- Parallel data collection: Collecting PK and ADA data concurrently can provide a more complete picture of the drug's performance.
However, multiplexing adds complexity. The monitoring plan must account for potential interference between analytes to preserve the sensitivity of the ADA detection.
Best Practices for Long-Term Maintenance
Avoiding costly mid-trial corrections requires a disciplined approach to assay maintenance. Best practices include:
- Regular Revalidation: Periodic checks of the assay's key parameters (e.g., sensitivity and cut point) confirm that the method remains suitable for its intended use.
- Incurred Sample Reanalysis (ISR): While typically associated with PK, reanalyzing a portion of study samples for ADA can confirm the method's reproducibility over time.
- Real-time Data Review: analyzing data on a rolling basis rather than waiting until the end of the study allows for immediate corrective action.
Ensuring the validity of these methods is imperative to generate reliable, actionable data throughout the study lifecycle.
Conclusion
Ultimately, the purpose of rigorous monitoring of ADA assays is patient safety. Unwanted immune responses can alter a drug's pharmacokinetics or lead to adverse events. If the assay performance wavers, the ability to correlate clinical observations with immunogenicity status is lost. By maintaining strict oversight of the assay life cycle, bioanalytical teams protect the integrity of the data. This vigilance ensures that when the final clinical study report is filed, the conclusions drawn about the drug's safety and efficacy are based on reliable, high-quality evidence.













0 Comments